In Part One, I covered the following concepts:
Base manifestations of hope
Children: Hope and immortality
Human population levels
Compound effects of health and fertility
Autoimmunity
Degradations of the human immune system
Mutation
The limits of knowledge on choice
Credentialism’s effect on the validity of a hypothesis
Here in Part Two, I will introduce specific, separate components of the End of Hope hypothesis, and show you as best I can what I think are key indications or current states of knowledge about those components, within the context of the hypothesis.
These components relate directly to the concepts in Part One and are constituent parts of the End of Hope hypothesis. It is my hope that by structuring these articles in this way - concepts, components, hypothesis - readers with no other prior understanding or awareness may be just as able to evaluate the hypothesis as others.
I’ll warn you now: this is a beast of a section and, depending upon how your mind works, your reading preferences and your bent for science, you may find my presentation of these components or their nature highly off-putting because they are either complex, boring, seemingly disparate or I just lack the skill to write about them in a compelling way.
I am well aware of both the limits of my own knowledge and skill as a writer and these articles are challenging to write in a way that builds to my anticipated end goal.
I feel that it is important to show you these structured chunks before I show you what would be considered the narrative glue between them.
That narrative glue IS the hypothesis itself. I feel that in order for you to evaluate the validity of the hypothesis and judge its merits both linguistically and to some degree technically, I have to show you all of these concepts and components first, before I show you the hypothesis. There is a final, key reason why the hypothesis comes last, which I will explain when appropriate.
In what follows, LINE-1 is is probably the toughest bit. I reckon many will not make it past that point because of either the topic’s inherent complexity, my inability to fairly and accurately explain LINE-1, and/or because I cannot provide definite answers to the questions posed that this throws up.
There aren’t meant to be definite answers, because this is a component in an untested hypothesis. You must be able to tolerate ambiguity borne out of our own lack of knowledge and understanding in order to evaluate the End of Hope hypothesis itself.
Accept, like I do, that unless you are skilled and knowledgeable in this field, you will not fully know or understand what follows on LINE-1. What you, like I, can derive from what follows is a sense of the complex dynamism of genetics as well as the limits of our understanding of it. From there you may, as I do, be left with senses of wonder and caution. It is important to be able to attempt the LINE-1 material here then see it in the context of the hypothesis so that you may evaluate that part of the hypothesis. If you have the knowledge or skill, the way LINE-1 features in the hypothesis may itself completely invalidate the hypothesis.
What I am looking for, initially, is validation of the hypothesis i.e. is it feasible and does it hang together?
If it doesn’t, then good, we can all go back to bed. If it does, then the next question is this:
If the hypothesis is valid, what investigational priority does it deserve?
There is a reason why I have called it the End of Hope hypothesis, which you will probably have worked out just from Part One.
Hypothesis components
Fertility data
People have been looking at birth rate signals. Jikkyleaks, Igor Chudov and Josh Guetzkow are just a few. You can find recent posts by starting here, with Chudov’s article. I’ve included PDFs here of some of the German material for preservation.
Note: Chudov’s article contains links to birth rate data for Canada, Sweden, Taiwan, Hungary, Germany, North Dakota, UK and Switzerland.
Rate decreases they report range from -10% to -28%, with some claims of event correlation.
Both Chudov and Jikkyleaks were suspended from Twitter shortly after they posted about this topic.
Here is Dóra Dúró flagging the issue of declining birth rates in the Hungarian parliament.
Autoimmunity
The UK’s MHRA states the following in respect of the mechanism of action of the Covid-19 mRNA gene therapies:
“After injection, the lipid nanoparticles (LNP) are taken up by the cells, and the RNA is released into the cytosol. In the cytosol, the RNA is translated into the encoded viral protein. The viral spike (S) protein antigen induces an adaptive immune response through neutralising antibodies. Furthermore, as the expressed spike (S) protein is being degraded intracellularly, the resulting peptides can be presented at the cell surface, triggering a specific T cell-mediated immune response with activity against the virus and infected cells.”
This mechanism of action also applies in part to DNA-based vaccines from AZ and J&J. All Covid-19 vaccines induce cellular production and expression of Spike protein. The key difference between DNA and mRNA vaccines is that the DNA payload actually enters your cell nucleus and triggers gene expression of mRNA, which then leaves the nucleus for translation by the ribosome in the cell’s cytosol. mRNA vaccines directly deliver their synthetic mRNA payload into the cytosol for translation and shouldn’t enter the nucleus and don’t need to trigger gene expression in the cell nucleus.
Three primary outcomes follow, as per the MHRA’s explicit statement:
Spike protein is produced in the cell;
Spike protein is expressed by the cell, some of which may be free-floating;
Spike protein and/or related materials e.g. peptides, may present at the cell surface.
All three of these outcomes can trigger an immune response that can lead to the destruction of the cell in question because all three of those outcomes can alert the immune system to what looks like infection of the cell through the presence of the foreign Spike protein antigen. Further, Spike or foreign peptides presenting at the cell membrane can mark the entire cell as foreign or infected, triggering a destructive T-cell mediated immune response that kills the whole cell and everything in it.
This is what the MHRA is describing. That quote relates only to Covid-19 vaccination with Pfizer’s BNT162b2, not SARS-CoV-2 infection. Note that the MHRA uses the term “infected cells”. Think about why that term is mechanistically appropriate.
If you were infected by SC2 and the virus replicated inside your cells, Spike proteins would be produced inside the cell as a function of the viral reproduction process occurring in the cell that the virus has infected. The new virus particles would be expressed by the cell or escape, destructively or non-destructively. Thus, throughout this process, Spike protein is being expressed. As far as Spike protein is concerned, both viral infection and vaccination with Covid-19 “vaccines” has the same outcome. Cells in a vaccinated person behave as though they are virally infected in that they produce and express Spike protein. That, in the case of viral infection, those Spike proteins are attached to an instance of the virus, is immaterial. Spike antigens are being produced and expressed in both cases and are detectable by the immune system.
In the case of viral infection, your immune response deals with the virus and your infected cells. I have already argued that this is not “autoimmunity” but the correct, desired natural adaptive immune response, where:
an adaptive immune response from vaccination could, effectively, result in autoimmunity in the host, and where “these responses are destructive, it is crucial that they be made only in response to molecules that are foreign to the host and not to the molecules of the host itself. The ability to distinguish what is foreign from what is self in this way is a fundamental feature of the adaptive immune system.” (Molecular Biology of the Cell, 4th Edition).
So far, so semi-theoretical, right?
The International Journal of Molecular Sciences published: “Intramyocardial Inflammation after COVID-19 Vaccination: An Endomyocardial Biopsy-Proven Case Series” 1.
“The present study summarizes EMB-based diagnostics of 15 patients with clinical suspicion of myocarditis following vaccination against SARS-CoV-2. It identifies 14 of 15 patients with myocardial inflammation, ranging from inflammatory cardiomyopathy to active myocarditis and severe giant cell myocarditis. Although a causal relationship between vaccination and the occurrence of myocardial inflammation cannot be established based on the findings, the cardiac detection of spike protein, the CD4+ T-cell-dominated inflammation and the close temporal relationship argue for a vaccine-triggered autoimmune reaction.”
This is just one third party study that points to a possibility of vaccine-triggered autoimmune reaction, but the MHRA has already admitted that destructive immune responses are, by their design, induced by Covid-19 gene therapies.
Now ask yourself how many of the known and suspected adverse effects of Covid gene therapies could be due to autoimmune reactions that involve cellular damage?
Where are the known and suspected adverse effects occuring? Anywhere. Everywhere. So they can’t be triggered by the gene therapy, which is injected into the deltoid and only dealt with there and in the local lymphatic system, as per the general understanding of vaccines.
Biodistribution
mRNA Covid gene therapies can go anywhere and everywhere in the body. This is admitted and proven by the manufacturers, the MHRA, EMA, FDA etc, and they cross the blood brain barrier. Therefore, they could act in any tissue or system, causing local damage and wider effects, including inducing autoimmunity anywhere.
“Information regarding the potential distribution of the test articles to sites other than the injection site following IM administration has been provided and is under review as part of the ongoing rolling assessment.”
(MHRA, Summary of the Public Assessment Report for COVID-19 Vaccine Pfizer/BioNTech, 24/05/2022)
The EMA EPAR document of 11/03/21 stated:
“mRNA could be detected in all examined tissues except the kidney. This included heart, lung, testis and also brain tissues, indicating that the mRNA/LNP platform crossed the blood/brain barrier.”
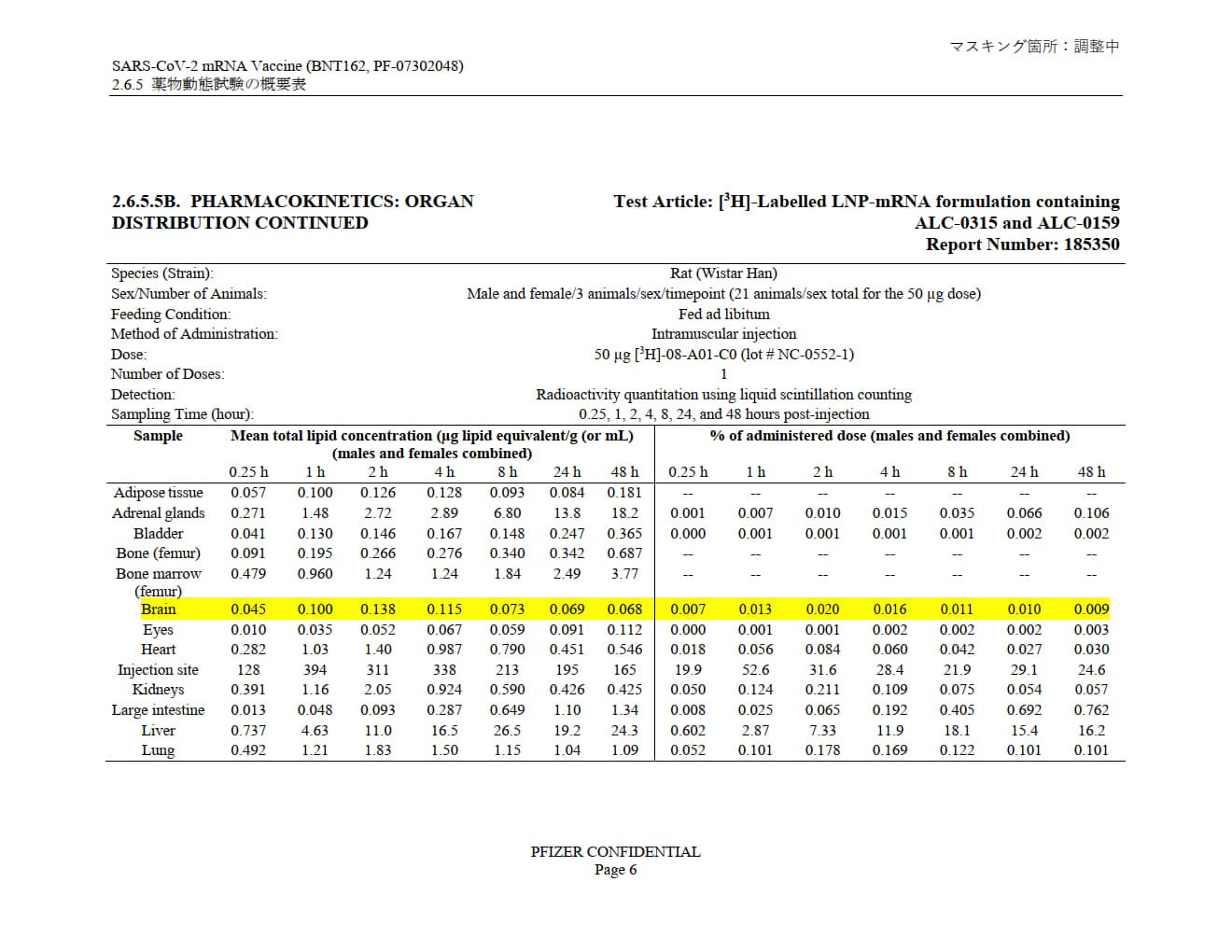
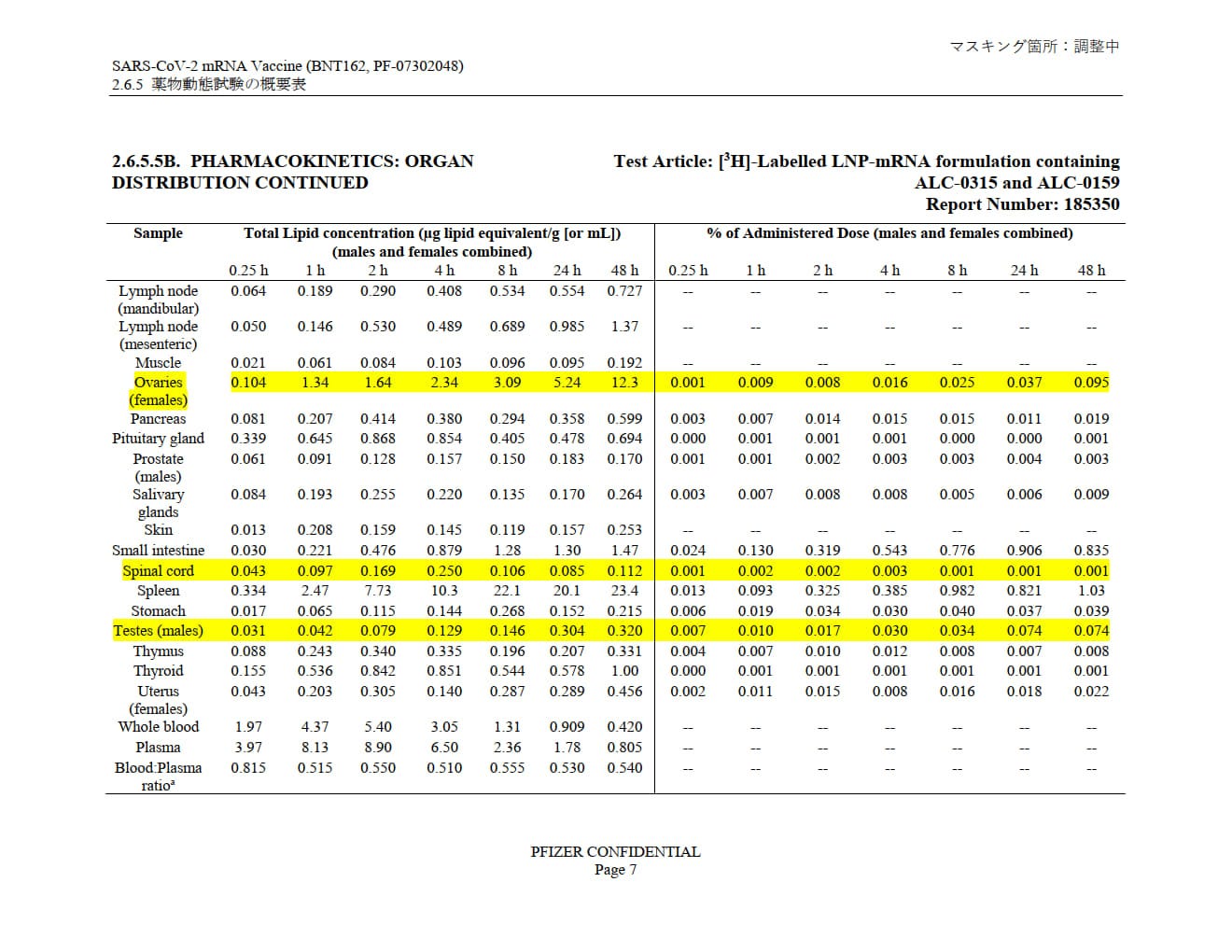
Pfizer's own pharmacokinetic study, “SARS-CoV-2 mRNA Vaccine (BNT162, PF-07302048) 2.6.4 薬物動態試験の概要文 (translation: “Summary of pharmacokinetic study”)” provided to the Japanese government shows LNP distribution through practically the entire body, with accumulation in many tissues, systems and organs over the 48-hour study window.
Note: in all the highlighted organs, over 48 hours the LNPs build up. They build by 12x in the ovaries and 10x in the testes.
Genotoxicity, Mutagenicity & Carcinogenicity
Genotoxicity & Mutagenicity are inter-related.
Genotoxicity is the property of chemical agents that damage the genetic information within a cell causing mutations, which may lead to cancer. While genotoxicity is often confused with mutagenicity, all mutagens are genotoxic, but some genotoxic substances are not mutagenic. The alteration can have direct or indirect effects on the DNA: the induction of mutations, mistimed event activation, and direct DNA damage leading to mutations. The permanent, heritable changes can affect either somatic cells of the organism or germ cells to be passed on to future generations.[1] Cells prevent expression of the genotoxic mutation by either DNA repair or apoptosis; however, the damage may not always be fixed leading to mutagenesis.
https://en.wikipedia.org/wiki/Genotoxicity
In genetics, a mutagen is a physical or chemical agent that permanently changes genetic material, usually DNA, in an organism and thus increases the frequency of mutations above the natural background level. As many mutations can cause cancer in animals, such mutagens can therefore be carcinogens, although not all necessarily are. All mutagens have characteristic mutational signatures with some chemicals becoming mutagenic through cellular processes.
The process of DNA becoming modified is called mutagenesis. Not all mutations are caused by mutagens: so-called "spontaneous mutations" occur due to spontaneous hydrolysis, errors in DNA replication, repair and recombination.
If you wanted to fully determine whether something was a human genotoxin and/or mutagen, what level of testing would you accept and in what organisms? Would you demand full scale in vitro, animal and in vivo testing for both before making a determination? If you had not done this level of testing would it be reasonable for you to state that the thing in question was not genotoxic or mutagenic to humans? Or would you have to say something else?
If a product was comprised of multiple components, would your testing involve testing of each individual component and the final product, to the same standard and degree, in order to determine that ingredients in isolation and in combination did not demonstrate genotoxicity or mutagenicity?
Would your testing standards change depending upon the nature of the product and if so, to what degree? Consider three examples: a shoe, a drug and a vaccine.
As for carcinogenisis:
Carcinogenesis, also called oncogenesis or tumorigenesis, is the formation of a cancer, whereby normal cells are transformed into cancer cells. The process is characterized by changes at the cellular, genetic, and epigenetic levels and abnormal cell division… According to the prevailing accepted theory of carcinogenesis, the somatic mutation theory, mutations in DNA and epimutations that lead to cancer disrupt these orderly processes by interfering with the programming regulating the processes, upsetting the normal balance between proliferation and cell death.[1][2][3][4][5] This results in uncontrolled cell division and the evolution of those cells by natural selection in the body. Only certain mutations lead to cancer whereas the majority of mutations do not.[citation needed]
Variants of inherited genes may predispose individuals to cancer. In addition, environmental factors such as carcinogens and radiation cause mutations that may contribute to the development of cancer.
According to the MHRA, in the case of the AZ “vaccine”:
Genotoxicity
No genotoxicity studies were performed.
Carcinogenicity
No carcinogenicity studies were performed. Carcinogenicity testing is generally not considered necessary to support the development and licensure of vaccine products for infectious diseases (WHO, 2005).
At the point the MHRA granted Conditional Marketing Authority to the AZ “vaccine”, it did not know whether it was genotoxic because no testing had been performed. No explanation as to why not is given in the MHRA’s EPAR document.
The MHRA also does not know whether the “vaccine” is carcinogenic. This is because per a WHO statement, if something is called a “vaccine”, there is a prevailing assumption that no one should check if it is carcinogenic, irrespective of what the “vaccine” actually is, how it works or what its ingredients are.
According to the EMA, in respect of Moderna’s Spikevax, some genotoxicity studies were conducted on the SM-102 ingredient alone, which returned no concerns, and in final formulation. “The other lipid components contained in the final formulation, i.e. PEG2000-DMG, DSPC and cholesterol, were not separately tested but are contained in the formulation tested in the in vivo genotoxicity studies, which is acceptable. DSPC and cholesterol do not raise any concern in terms of genotoxic potential.” Within the scope of the testing and the extent to which it is described, EMA states that no genotoxic effects we seen.
No carcinogenicity studies were submitted. This is scientifically acceptable and in line with relevant guidelines on non-clinical development of vaccine candidates. The components of the vaccine formulation are lipids and natural nucleosides that are not expected to have carcinogenic potential.
I leave it to you to research the regulators’ write ups on genotoxicity, mutagenicity and carcinogenicity across the full range of Covid gene therapies.
CYP19A1 gene
This gene does important things. Here’s an overview:
The CYP19A1 gene provides instructions for making an enzyme called aromatase. This enzyme converts a class of hormones called androgens, which are involved in male sexual development, to different forms of the female sex hormone estrogen.
In cells, aromatase is found in a structure called the endoplasmic reticulum, which is involved in protein production, processing, and transport. The activity (expression) of aromatase varies among different cell types depending on the cells' need for estrogen. In females, aromatase is most active in the ovaries, where it guides sexual development. In males, aromatase is most active in fat (adipose) tissue. In both males and females, estrogen plays a role in regulating bone growth and blood sugar levels. During fetal development, aromatase converts androgens to estrogens in the placenta, which is the link between the mother's blood supply and the fetus. This conversion in the placenta prevents androgens from directing sexual development in female fetuses. After birth, the conversion of androgens to estrogens takes place in multiple tissues.
The enzyme that the CYP19A1 gene makes - aromatase - is found in the endoplasmic reticulum, which is a structure inside every cell that has a nucleus (eukaryotic cells), which are most of the cells in the human body. Aromatase is most active in female ovaries and male fat tissue. Screw with the CYP19A1 gene and aromatase deficiency can occur where “abnormal hormone levels lead to impaired sexual development in affected females and unusual bone growth, insulin resistance, and other signs and symptoms in both males and females with the condition,” or an excess can also occur where “affected males have enlarged breasts (gynecomastia) and short stature; affected females can have irregular menstrual periods and short stature.”
CDKN1B gene
The CDKN1B gene provides instructions for making a protein called p27. This protein is found in cells and tissues throughout the body. Within cells, p27 is located primarily in the nucleus, where it plays a critical role in controlling cell growth and division. It helps regulate the cell cycle, which is the cell's way of replicating itself in an organized, step-by-step fashion. Specifically, p27 normally blocks cells from entering the phase of the cell cycle when DNA is copied (replicated) in preparation for cell division. By blocking cell cycle progression, p27 prevents cells from dividing too quickly or at the wrong time. Based on this function, p27 is described as a tumor suppressor protein. Studies suggest that p27 is also involved in controlling cell differentiation, which is the process by which cells mature to carry out specific functions.
Because p27 plays such a key role in controlling cell division, its activity is tightly regulated. Regulation can occur through modification of the p27 protein's structure, its interaction with other proteins, or its localization within the cell. For example, when p27 is held (sequestered) in the fluid that surrounds the nucleus (the cytoplasm) instead of being transported into the nucleus, the protein is unavailable to block cell cycle progression. Researchers believe that p27 may have other functions in the cytoplasm, but these functions have not been well described.
Damage to this gene can lead to endocrine problems.
Failure of the endocrine system means that your body is unable to properly monitor and modulate hormone levels, via which cells and systems communicate with each other, giving rise to major health issues.
Reverse transcription of Covid-19 BNT162b2 and SARS-CoV-2 RNA
Research has shown, some time ago that both the mRNA Pfizer “vaccine” and SC2 itself may both be able to integrate with the human genome under certain circumstances and in certain cell lines/types. According to manufacturers and regulators, this could not happen.
However, this capability is could have been found had suitable genotoxicity, mutagenicity and carcinogenicity testing been carried out.
Reverse transcription (which occurs in both prokaryotes and eukaryotes) is the synthesis of DNA from an RNA template. A class of RNA viruses, called retroviruses, are characterized by the presence of an RNA‐dependent DNA polymerase (reverse transcriptase). The virus that causes AIDS, Human Immunodeficiency Virus (HIV), is a retrovirus. Because nuclear cell division doesn't use reverse transcriptase, the most effective anti‐HIV drugs target reverse transcriptase, either its synthesis or its activity. Telomerase, discussed in the previous section, is a specialized reverse transcriptase enzyme. See Figure 1.
Current Issues in Molecular Biology published “Intracellular Reverse Transcription of Pfizer BioNTech COVID-19 mRNA Vaccine BNT162b2 In Vitro in Human Liver Cell Line”1:
“The cell model that we used in this study is a carcinoma cell line, with active DNA replication which differs from non-dividing somatic cells. It has also been shown that Huh7 cells display significant different gene and protein expression including upregulated proteins involved in RNA metabolism [56]. However, cell proliferation is also active in several human tissues such as the bone marrow or basal layers of epithelia as well as during embryogenesis, and it is therefore necessary to examine the effect of BNT162b2 on genomic integrity under such conditions. Furthermore, effective retrotransposition of LINE-1 has also been reported in non-dividing and terminally differentiated cells, such as human neurons.
“Conclusions: Our study is the first in vitro study on the effect of COVID-19 mRNA vaccine BNT162b2 on human liver cell line. We present evidence on fast entry of BNT162b2 into the cells and subsequent intracellular reverse transcription of BNT162b2 mRNA into DNA.”
On December 13, 2020, research was published titled, “SARS-CoV-2 RNA reverse-transcribed and integrated into the human genome”,2 which stated:
“...we found chimeric transcripts consisting of viral fused to cellular sequences in published data sets of SARS-CoV-2 infected cultured cells and primary cells of patients, consistent with the transcription of viral sequences integrated into the genome. To experimentally corroborate the possibility of viral retro integration, we describe evidence that SARS-CoV-2 RNAs can be reverse transcribed in human cells by reverse transcriptase (RT) from LINE-1 elements or by HIV-1 RT, and that these DNA sequences can be integrated into the cell genome and subsequently be transcribed. Human endogenous LINE-1 expression was induced upon SARS-CoV-2 infection or by cytokine exposure in cultured cells, suggesting a molecular mechanism for SARS-CoV-2 retro-integration in patients. This novel feature of SARS-CoV-2 infection may explain why patients can continue to produce viral RNA after recovery and suggests a new aspect of RNA virus replication.”
LINE-1
What is LINE-1?
There is a lot of technical language and tough concepts involved here. I will first present my lay interpretation then excerpts from papers. In the context of the hypothesis, there’s only so much we need to know in order to judge the hypothesis.
LINE-1 stands for “long-interspersed nuclear elements-1”. They are DNA elements of the human (and other species) genome. They constitute up to maybe 46% of your whole genome. They are mobile and they can literally move around your DNA sequence and write in to it (transposition). Their discovery proved that human DNA is not fixed or static, but instead changes over time for internal and external reasons. The power and ability of LINE-1 to “jump” and in doing so change your DNA is tightly controlled throughout your lifespan. To move around and write into your DNA, LINE-1 must be able to perform a complete cycle of steps in specific order.
The completion of the human genome sequence has revealed the sheer abundance, diversity and importance of our transposons (Lander, 2001). Transposition is an ongoing process, actively changing the genome, occasionally for the worse(Kazazian and Moran, 1998; Ostertag and Kazazian, 2001a; Gilbert et al., 2002; Symer et al., 2002)
DNA transposition is the movement of a defined DNA segment (a transposon) from one genomic site to another. Mechanistically, there are two types of transpositions: (1) cut and paste transposition, and (2) replicative transposition.
https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/dna-transposition
Here’s a more technical version of the above. The source article is lengthy and hard work, but informative.
Transposable elements (TEs) are mobile DNA elements that represent almost half of the human genome. Transposition of TEs has been implicated as a source of genome evolution and acquisition of new traits but also as an origin of diseases. The activity of these elements is therefore tightly regulated during the life cycle of each individual, and many recent discoveries involved the genetic and epigenetic mechanisms in their control… one specific family of TEs: the long-interspersed nuclear elements-1 (LINE-1 or L1). LINE-1 elements are the most representative class of retrotransposons in mammalian genomes.
The popularity of these classes of DNA stems from their ability to translocate to a new location in a genome, explaining why TEs are also called ‘jumping genes’ or ‘mobile elements’. Moreover, TEs account for 46% of the human genome.
Active L1 elements must be able to perform a complete retrotransposition cycle that includes the following steps in chronological order: transcription of L1 RNA, export into the cytoplasm, translation of ORF1 and ORF2, association of L1 RNA with ORF1 and ORF2 proteins to form ribonucleoprotein (RNP) particles (11), return to the nucleus, reverse transcription, and integration at a new genomic location – a one-step process also called target primed reverse transcription (TPRT).
https://www.degruyter.com/document/doi/10.1515/bmc-2014-0018/html
What is LINE-1 involved in?
LINE-1 is an indicator of cancer and other diseases. It also modulates the immune system and affects how it works. If LINE-1 gets out of control, the innate immune response can be triggered, the immune system activated and autoimmunity and inflammation may result. So, conversely, limiting (downregulating) LINE-1 could be a way to treat some diseases e.g. inflammatory or immune diseases.
Long interspersed nuclear element-1 (LINE-1) retrotransposition is a major hallmark of cancer accompanied by global chromosomal instability, genomic instability, and genetic heterogeneity and has become one indicator for the occurrence, development, and poor prognosis of many diseases. LINE-1 also modulates the immune system and affects the immune microenvironment in a variety of ways. Aberrant expression of LINE-1 retrotransposon can provide strong stimuli for an innate immune response, activate the immune system, and induce autoimmunity and inflammation. Therefore, inhibition the activity of LINE-1 has become a potential treatment strategy for various diseases. In this review, we discussed the components and regulatory mechanisms involved with LINE-1, its correlations with disease and immunity, and multiple inhibitors of LINE-1, providing a new understanding of LINE-1.
In cancer cells, however, L1s are aberrantly activated and may have a role in genome instability, one of the hallmarks of cancer pathogenesis.
Because L1 expression is apparently activated (or upregulated) in some cancers, the L1-encoded proteins are also detectable.31 They may participate in tumorigenesis and their expression profiles can be of potential clinical significance. A study of breast cancer by Chen et al.,74 for example, revealed that the majority of invasive cancers expressed L1 proteins in the cytoplasm, with 28–31% of them showing nuclear expression. Moreover, patients with L1 nuclear expression suffered from more lymph node metastasis and worse prognosis relative to patients without. Given that nuclear ORF1p and ORF2p are building blocks for L1 retrotransposition, whether or not this prognostic association is attributable to retrotransposition-induced mutagenesis requires further investigation.
Is LINE-1 retrotransposition associated with the fertility and development cycles?
Yes. LINE-1 activities have been demonstrated in germ cells (sperm and eggs) that carry through to offspring, and also shown to occur during embryo development, although the research quoted below suggests the degree to which this may occur might be less than was previously thought.
LINE-1 retrotransposition events may more frequently occur in development rather than in germlines. LINE-1 activity in somatic cells (cells that are not sperm or egg cells) may have effects on the learning and behaviour of the person, as well as cancer development and non-inheritable diseases. LINE-1 RNA can also alter the DNA of offspring.
Together, these data indicate that L1 RNA transcribed in male or female germ cells can be carried over through fertilization and integrate during embryogenesis, an interesting example of heritability of RNA independent of its encoding DNA. Thus, L1 creates somatic mosaicism during mammalian development, suggesting a role for L1 in carcinogenesis and other disease.
Despite the tremendous impact of L1 on the human genome, much of the process of L1 retrotransposition in vivo remains unexplored. The several hundred thousand copies of L1 in the human genome demonstrate that they have accumulated over time.
Somatic retrotransposition events that do not get incorporated into germ cells are not heritable and will not accumulate in the genome. In addition, L1 RNA and proteins have been found predominantly in germ cells and infrequently in differentiated tissues (Branciforte and Martin 1994; Trelogan and Martin 1995). Therefore, the prevailing view has been that the bulk of L1 retrotransposition occurs in germ cells.
Here, we demonstrate that retrotransposition occurring directly in germ cells is uncommon. Most human and mouse L1 retrotransposition events occur in embryonic development; only a fraction of these events get into the germline and are heritable.
In our study, human and mouse L1 RNA was carried over through both male and female germ cells to the next generation.
It is of great interest that most human L1 retrotransposition events likely occur in development rather than in the germline, using L1 RNAs transcribed in the embryo and in developing germ cells. These data suggest a role for somatic L1 retrotransposition events in human development with potential effects on learning and behavior, as previously suggested (Muotri et al. 2005), carcinogenesis (Miki et al. 1992), and other conditions with somatic, nonheritable mutations in their etiology. That L1 RNA can alter the DNA of a subsequent generation is another example of the noncanonical role RNA can play in determining the human phenotype.
How can LINE-1 be affected or interfered with?
LINE-1 activity can be directly and indirectly increased (upregulated) or decreased (downregulated) by a variety of means including the use of drugs and the presence or absence of other genetic elements and proteins. LINE-1 activity is upregulated by the absence of some elements that therefore seem to act to suppress it. Also, by introducing the presence of DNA methyltransferase 3 alpha (an enzyme produced by the DNMT3A gene) LINE-1 expression levels can be upregulated. Thus, LINE-1 activity or expression can be modulated, possibly by deliberate external means.
It was found in breast cancer cells treated with paclitaxel that DNMT3a, a member of the DNA methyltransferase family, could enhance the methylation level in the gene by binding to the inner region of the LINE-1 gene, and then upregulate the expression level of LINE-1. Downregulating the expression of DNMT3a can effectively inhibit the expression of LINE-1 (Wang et al., 2020). LINE-1 retrotransposon silenced also through histone modifications. Histone demethylase KDM4B may enhance the LINE-1 retrotransposition efficacy, whereas depletion of KDM4B reduced it in breast cancer (Xiang et al., 2019). Elevated LINE-1 expression was found in PC9 drug-tolerant persister (DTP) cancer cells treated with the EGFR inhibitor erlotinib. HDAC inhibitors can suppress LINE-1 in DTP cancer cells (Guler et al., 2017). Currently, DNA methyltransferase inhibitors and histone deacetylase inhibitors have entered clinical trials (Gaillard et al., 2019).
Fanconi Anemia (FA) is a genetic disorder characterized by elevated cancer susceptibility and pro-inflammatory cytokine production. Using SLX4FANCP deficiency as a working model, we questioned the trigger for chronic inflammation in FA. We found that absence of SLX4 caused cytoplasmic DNA accumulation, including sequences deriving from active Long INterspersed Element-1 (LINE-1), triggering the cGAS-STING pathway to elicit interferon (IFN) expression. In agreement, absence of SLX4 leads to upregulated LINE-1 retrotransposition. Importantly, similar results were obtained with the FANCD2 upstream activator of SLX4. Furthermore, treatment of FA cells with the Tenofovir reverse transcriptase inhibitor (RTi), that prevents endogenous retrotransposition, decreased both accumulation of cytoplasmic DNA and pro-inflammatory signaling.
Interestingly, absence of DDR proteins can lead to upregulated retrotransposition (Stetson et al., 2008), which constitutes a potential source of immunogenic endogenous nucleic acids.
Altogether, our data demonstrate a correlation between absence of a functional FA DNA repair pathway, elevated pro-inflammatory cytokine production through activation of the cGAS-STING pathway (Fig. 2, Fig. 5) and upregulated LINE-1 activity (Fig. 4, Fig. 5).
In this model, we observed that absence of SLX4 leads to upregulated LINE-1 retrotransposition.
In addition, in SLX4-deficient cells, LINE-1 transcription and retrotransposition are increased and the nucleoplasm contains immunogenic nucleic acids that may subsequently be exported to the cytoplasm and recognized by the cGAS-STING pathway to induce pro-inflammatory cytokine production (Fig. 7).
It would thus be interesting to investigate the influence of SLX4-deficient cells-derived DNA on adaptive immune responses.
Can LINE-1 changes be inherited?
Yes. Line-1 changes to sperm or eggs can affect the embryo that results from their fusion during fertilisation and they can integrate in the embryo’s genome.
Together, these data indicate that L1 RNA transcribed in male or female germ cells can be carried over through fertilization and integrate during embryogenesis, an interesting example of heritability of RNA independent of its encoding DNA. Thus, L1 creates somatic mosaicism during mammalian development, suggesting a role for L1 in carcinogenesis and other disease.
That you made it this far is a demonstration of rare qualities.
Part Three will bring this altogether.
Covid: The End of Hope Hypothesis (part three)
Ways to validate the hypothesis There are three ways to validate the hypothesis, two of which could be considered non-technical or non-specialist. Linguistic validation: do the terms used in the hypothesis make sense in context and are they sufficiently correctly or coherently used? For example, “discombobulation of LINE-1 phagocytes leading to reverse m…





If it was only mRNA and spike protein, the genocide would be easy to prove.
However, the injections contain the combination of a few dozen parasites, toxins, pathogens, and nanotech that interact with 5G and with each other, presenting virtually infinite ways to die.
https://rayhorvaththesource.substack.com/p/evidence-that-covid-symptoms-came
https://rayhorvaththesource.substack.com/p/a-few-more-sources-of-poisoning